-
 Eurex Energy
Eurex EnergyChemical recycling
by pyrolysisChemical recycling by pyrolysis: under normal atmospheric conditions without the participation of oxygen thermal processes cause the decomposition of the plastic polymer chain into smaller parts, where arises recycled / a mixture of liquid hydrocarbons, which is then used as a raw material for the petrochemical industry to produce new organic products or as a substitute for liquid fuels obtained by oil refining.
What are the benefits of chemical recycling Eurex Eco-01
-
The facility does not burden the environment with a large amount of exhaust fumes and greenhouse gases, on the contrary, there is also a significant reduction in the carbon footprint that would arise during the extraction of oil and its primary processing
-
our technology can make a significant contribution to the treatment of difficult-to-recycle plastic waste, and the recycled output serves as a substitute for primary fossil raw materials in the production process of new plastic products.
-
the use of final products in chemical recycling is incomparably greater than in mechanical recycling (95 % : 10 % ).
-
a significant part of mixed plastic waste cannot be mechanically recycled. Recycling efficiency is 95 % : 50 % in favor of chemical recycling.
The process of chemical recycling is practically self-sufficient in energy. External energy is only required by the process to drive electrical equipment.
-
-
 Pyrolysis – an established technology finds a new application
Pyrolysis – an established technology finds a new applicationPyrolysis
a little aboutIn the pyrolysis process, plastics are broken down into a range of basic hydrocarbons by heating in the absence of oxygen, or ‘cracking’ (sometimes referred to as thermal cracking). By utilising a distillation process, the hydrocarbon vapour can then be made into products ranging from heavy wax and oils to light oils and gas. It is possible to skew the production from heavier to lighter by adjusting process time and temperature. Heavier output products can also be reintroduced into the process for additional cracking into lighter products.
Pyrolysis products can be processed in much the same way as oil, using conventional refining technologies to produce building blocks for polymers. Alternatively, they can be used directly as a fuel.
Using pyrolysis to make feedstock for polyethylene and polypropylene production could fill a large processing gap as polyethylene and polypropylene cannot be depolymerised directly into monomers. Further, the plastic produced would be virgin-quality polymers and could be used in all the same applications (e.g., food packaging).
Pyrolysis production can be enhanced using catalytic degradation, where a suitable catalyst is used to promote the cracking reaction. The presence of a catalyst allows reaction temperature and time to be lowered. The process results in a much narrower product distribution of carbon atom number and increases lighter hydrocarbon production. This helps to increase the proportion of the output product for use in making more plastics.
Target Feedstock
- Polyolefins [Polyethylene (PE), Polypropylene (PP), Polybutylene (PB)]
- Polystyrene (PS)
- PMMA (poly-methymethacrylate) - acrylic glass
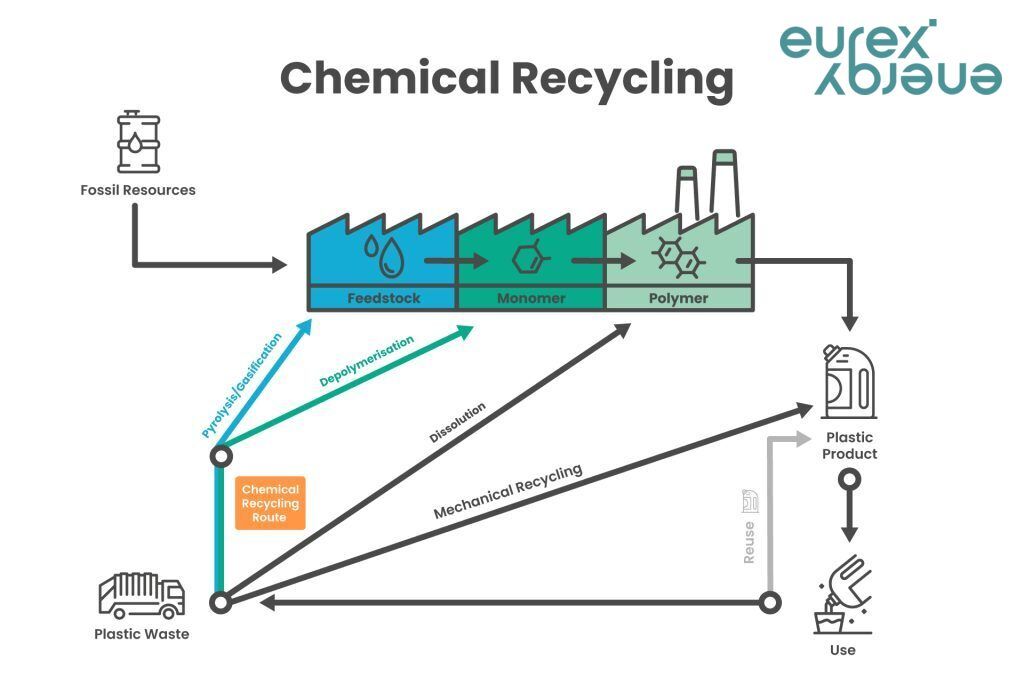
Chemical recycling
... is the process of converting polymeric waste by changing its chemical structure and turning it back into substances that can be used as raw materials for the manufacturing of plastics or other products. There are different chemical recycling technologies, e.g. pyrolysis, gasification, hydro-cracking and depolymerisation.

What are the benefits of chemical recycling?
Chemical recycling complements other plastic recycling options like mechanical, dissolution and organic recycling. Since it can deal with complex plastic waste streams, like films or laminates, chemical recycling can be used for plastic waste, which would otherwise result in incineration or landfill. With 67.5% of post-consumer plastic waste going to landfill and energy recovery across Europe, there is a clear potential for improvement.
Because chemical recycling breaks down polymers into their building blocks, it also allows the production of recycled plastic (recyclate) with virgin plastic properties that can be used in demanding applications, such as food contact.
The European Commission has set very ambitious circularity objectives for plastics. Regulatory targets of the recently revised waste directives are 10% max landfilling of municipal waste by 2035, 50% recycling of plastic packaging by 2025 and 55% by 2030. In this context, chemical recycling represents a positive step towards reducing disposed waste and contributing to a circular economy for plastics. There is also a huge potential for new jobs as the sector develops.
Significant investments in recycling technology
We have seen a significant increase in planned chemical recycling investment: from EUR 2.6 billion in 2025 to EUR 7.2 billion in 2030. The production of recycled plastics is estimated to increase to 1.2 Mt in 2025 and 3.4 Mt in 2030. With this planned contribution of 1.2 Mt of recycled plastics produced through chemical recycling by 2025, Plastics Europe plays a leading role in delivering on the European Commission’s Circular Plastics Alliance target of 10 Mt recycled plastics used in European products by 2025. Conversion to feedstock technologies (pyrolysis, gasification) represents 80% of the planned capacities.
Technology
ABOUT US
we are on social NETWORK
Eurex Energy, s.r.o. capitalized on its own development using foreign knowledge and experience in the production of new technology in the use of renewable and alternative energy sources, the EUREX ECO 01 equipment for the recovery of mixed waste plastics in the form of chemical recycling.ed.



